Exactly what are Illuminated Gun Scopes?
Atoms, molecules, aspects, isotopes... DRAB! Well, perhaps it is for some people, although personally, I believe it's rather fascinating. For one thing, everything on the globe is made from these kinds of invisible debris. Although it may seem of atoms when visualizing the smallest "thing" in the world, when ever asking yourself what an atom is made of, it becomes clear you will discover smaller dirt.
What is an Atom?
Atoms are made out of a combination of electrons, protons, and neutrons - subatomic particles. Just how many of these three subatomic debris an atom contains is determined by what element element it belongs to. Atoms are identified by the exclusive number of protons within it has the nucleus -- this is its atomic multitude. A stable atom must have an equal number of protons and bad particals.
Protons have a positive electric power charge whilst electrons have a negative a single. Therefore , in the event that there are additional protons than electrons, you have a absolutely charged ion, also known as an important cation. Conversely, if you have more electrons than protons, you have a in a wrong way charged ion, also known as a great anion. Atoms with a world wide web electrical request as referred to can be manufactured so unnaturally from your neutral express by ionizing radiation.
So we protected protons and electrons, but you may be wondering what does the neutron do? Good, you can think of neutrons as the stuff that binds the protons together. As to why do they have to be locked together? As stated above, protons and electrons will be electrically recharged, and as such, will naturally repel dirt of the same signal. This is why groups of protons have to have neutrons to support them mutually. Hydrogen-1 will not have any kind of neutrons since it only offers one proton.
What is an Isotope?
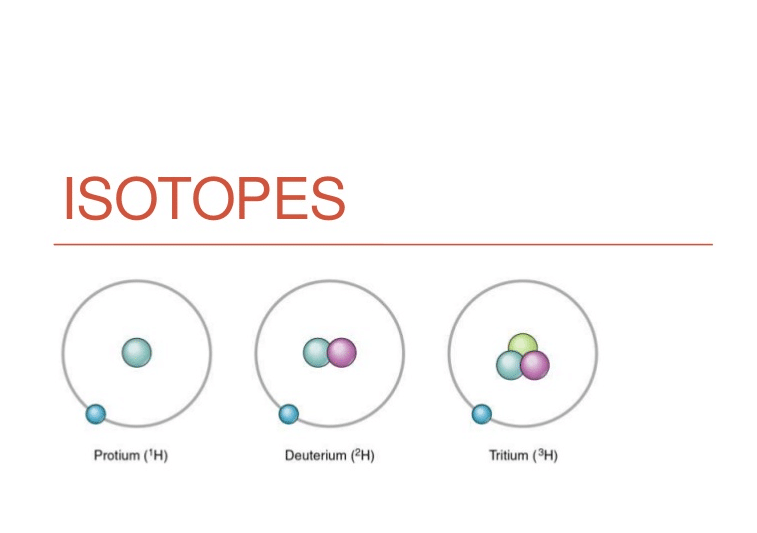
Therefore we know that the atomic number is derived from how many protons in an atom's nucleus, but what about isotopes? An isotope is usually defined through number of neutrons in an atom's nucleus. In a given element element, there are often a number of these isotopes. For instance , hydrogen provides 1 proton, but according to what hydrogen isotope it really is, the number of neutrons vary.
https://higheducationhere.com/isotopes/ are referred to as by their presented chemical ingredient, followed by their atomic mass, as in hydrogen-1, hydrogen-2, etc. This means that even though the number of protons remains consistent, the number of neutrons changes. Therefore hydrogen-2 could have a neutron as well as a proton, while hydrogen-3 will have two neutrons and a proton. Hydrogen-1 has only 1 wasserstoffion (positiv) (fachsprachlich) and no neutrons.
By subtracting the atomic number in the mass number, you discover the number of neutrons. Isotopes may be recognized on paper by a central part name accompanied by a distinct majority number that include hydrogen-3 as well as iodine-131. Every time speaking of various isotopes, radioactive or not, they will be recognized by name. Therefore , familiarizing yourself while using structure from atoms and related language can be helpful.

What is an Atom?
Atoms are made out of a combination of electrons, protons, and neutrons - subatomic particles. Just how many of these three subatomic debris an atom contains is determined by what element element it belongs to. Atoms are identified by the exclusive number of protons within it has the nucleus -- this is its atomic multitude. A stable atom must have an equal number of protons and bad particals.
Protons have a positive electric power charge whilst electrons have a negative a single. Therefore , in the event that there are additional protons than electrons, you have a absolutely charged ion, also known as an important cation. Conversely, if you have more electrons than protons, you have a in a wrong way charged ion, also known as a great anion. Atoms with a world wide web electrical request as referred to can be manufactured so unnaturally from your neutral express by ionizing radiation.
So we protected protons and electrons, but you may be wondering what does the neutron do? Good, you can think of neutrons as the stuff that binds the protons together. As to why do they have to be locked together? As stated above, protons and electrons will be electrically recharged, and as such, will naturally repel dirt of the same signal. This is why groups of protons have to have neutrons to support them mutually. Hydrogen-1 will not have any kind of neutrons since it only offers one proton.
What is an Isotope?
Therefore we know that the atomic number is derived from how many protons in an atom's nucleus, but what about isotopes? An isotope is usually defined through number of neutrons in an atom's nucleus. In a given element element, there are often a number of these isotopes. For instance , hydrogen provides 1 proton, but according to what hydrogen isotope it really is, the number of neutrons vary.
https://higheducationhere.com/isotopes/ are referred to as by their presented chemical ingredient, followed by their atomic mass, as in hydrogen-1, hydrogen-2, etc. This means that even though the number of protons remains consistent, the number of neutrons changes. Therefore hydrogen-2 could have a neutron as well as a proton, while hydrogen-3 will have two neutrons and a proton. Hydrogen-1 has only 1 wasserstoffion (positiv) (fachsprachlich) and no neutrons.
By subtracting the atomic number in the mass number, you discover the number of neutrons. Isotopes may be recognized on paper by a central part name accompanied by a distinct majority number that include hydrogen-3 as well as iodine-131. Every time speaking of various isotopes, radioactive or not, they will be recognized by name. Therefore , familiarizing yourself while using structure from atoms and related language can be helpful.
Public Last updated: 2022-01-09 04:24:28 PM
